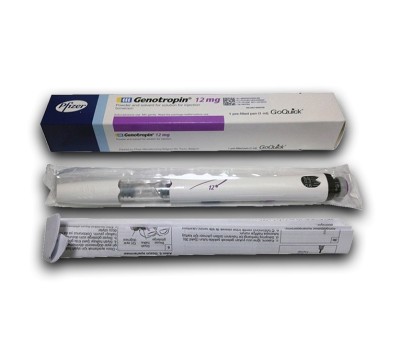
Genotropin QuickPen 36i.u. (12mg)
| Pack: | Price per Item | Total |
| $360.00 | $360.00 | |
| $350.00 | $1 750.00 | |
| $340.00 | $3 400.00 |
| Dosage | 36iu (12MG) |
| Total amount | 1 Quick Pen |
| Component | Human Growth Hormone |
Buy Genotropin QuickPen 36 IU online on domestic-supply.com
Genotropin QuickPen 36 IU is a prefilled, multi-dose injectable pen containing recombinant human growth hormone (somatropin). Manufactured by Pfizer, Genotropin is a branded hgh formulation supplied in a 12mg (36 iu) delivery device designed for controlled subcutaneous administration. Somatropin is a synthetic version of naturally occurring human growth hormone produced by the pituitary gland. It is classified as an injectable peptide hormone and is typically prescribed in regulated medical settings for growth hormone deficiency and other clinically diagnosed endocrine conditions.
The QuickPen delivery system is designed to provide measured dose administration using a dial-based pen mechanism to support accuracy and consistency. As a biologic medication, somatropin functions by interacting with growth hormone receptors in various tissues, supporting cellular growth, protein synthesis, and metabolic regulation under medical supervision.
Genotropin products are manufactured according to established pharmaceutical standards. Individuals seeking to buy Genotropin should ensure proper medical evaluation and regulatory approval before ordering. Use of this medication should occur only under the direction of a licensed healthcare provider. Packaging, shipping, and handling should follow appropriate pharmaceutical storage guidelines to maintain approve product integrity.
Key Features
-
Active Ingredient: Somatropin (recombinant human growth hormone)
-
Dosage Strength: 36 IU (12 mg) per prefilled pen
-
Form: Multi-dose prefilled injectable pen for subcutaneous administration
-
Manufacturer: Pfizer (brand-origin product)
-
Packaging: Single QuickPen device with replaceable needle compatibility
-
Classification: Injectable peptide hormone (growth hormone analogue)
-
Delivery System: Dial-adjustable pen mechanism for measured dosing
-
Manufacturing Standards: Produced under regulated pharmaceutical manufacturing practices
-
Intended Use Context: Prescribed medical therapy for clinically diagnosed growth hormone-related conditions
Frequently Asked Questions (FAQs)
-
What is Genotropin QuickPen 36 IU?
Genotropin QuickPen 36 IU is a branded formulation of recombinant human growth hormone (somatropin) supplied in a prefilled injection pen. It is designed for controlled subcutaneous administration in medically supervised treatment programs addressing growth hormone deficiencies and related endocrine conditions. -
How does Genotropin work?
Somatropin acts by binding to growth hormone receptors in tissues throughout the body. This interaction supports cellular growth, protein synthesis, metabolic regulation, and tissue development. The mechanism mirrors the activity of endogenous growth hormone when administered under appropriate clinical supervision. -
What are general dosing considerations?
Dosing is individualized and determined by a qualified healthcare provider based on diagnosis, patient age, body weight, and treatment goals. It is not appropriate to self-determine dosage or frequency. All administration protocols should follow licensed medical guidance and applicable regulatory standards. -
What are potential side effects?
Possible side effects associated with somatropin may include injection-site reactions, fluid retention, joint discomfort, headache, or changes in glucose metabolism. Adverse effects vary between individuals. Any unusual symptoms should be discussed with a healthcare professional for proper evaluation. -
How should Genotropin be stored and handled?
Genotropin QuickPen should be stored under refrigeration as indicated in the product packaging. It should not be frozen or exposed to excessive heat. Proper needle handling and disposal procedures should be followed according to local healthcare regulations. -
How can authenticity be verified?
Authenticity can be confirmed by examining manufacturer labeling, packaging integrity, batch numbers, and official security features. Products should be sourced only from licensed pharmaceutical suppliers to reduce the risk of counterfeit or improperly stored medication.
Customer Reviews
Michael R. — Rating (4.6/5)
Packaging arrived intact and well-protected. The pen mechanism feels precise and professionally manufactured. Overall presentation matches what I would expect from a regulated pharmaceutical product.
Daniel H. — Rating (4.4/5)
Delivery was discreet and timely. The product labeling and batch information were clearly printed. Everything appeared consistent with authentic branded packaging.
Anthony L. — Rating (4.7/5)
The QuickPen system is straightforward in design. Build quality feels durable and reliable. Instructions were clear and professionally presented.
Steven M. — Rating (4.5/5)
Product arrived within the expected timeframe. Storage instructions were clearly included, and packaging integrity was maintained throughout transit.
Carlos T. — Rating (4.6/5)
Consistent presentation and secure packaging. Manufacturer information and dosage labeling were easy to verify. Overall experience was smooth and professional.